LiteAire Additional Product Information
Innovative Dual-Valved System
One-way inhalation valve:
- Clear valve located inside the mouthpiece
- Opens when patient inhales to provide a direct path for aerosolized drug to flow from chamber into mouthpiece
- Closes against valve seat when patient exhales to redirect exhaled breath from entering the chamber
One-way exhalation valve:
- Small valve located on outside of the bottom panel of LiteAire
- Opens when patient exhales allowing exhaled breath to flow out
- Closes when patient inhales
Both valves:
- Low resistance allowing patient to breathe normally through the LiteAire
- Unnecessary to remove device from mouth to exhale
Chamber Volume:
- LiteAire holding chamber volume is approximately 184 mL
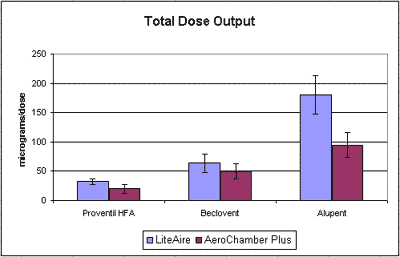
Use and Performance with Most MDIs
- The LiteAire enables the MDI drug particle sizes to remain concentrated toward the “respirable” 1-5 micron range
- May be used with most prescribed pMDI drugs
- MDI opening holds actuator firmly in an upright position